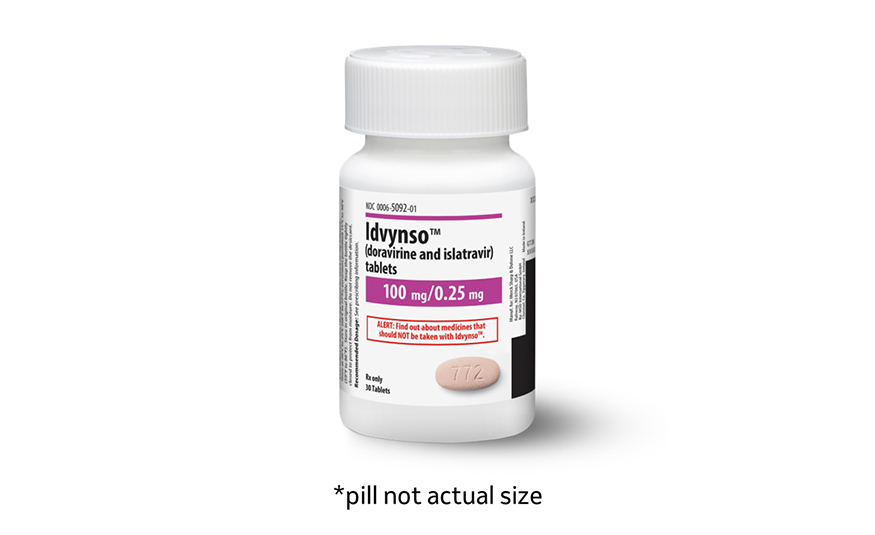
Idvynso combines two medicines (doravirine and islatravir) into a single tablet for adults whose HIV-1 is already under control. - PROVIDED BY MERCK

Idvynso combines two medicines (doravirine and islatravir) into a single tablet for adults whose HIV-1 is already under control. - PROVIDED BY MERCK
Merck wins FDA approval for once-daily HIV pill Idvynso
Kimberly Redmond//April 23, 2026//
Rahway-based Merck has secured U.S. Food and Drug Administration approval for its once-daily pill for HIV infections.
Branded as Idvynso, the treatment combines two medicines (doravirine and islatravir) into a single tablet for adults whose HIV-1 is already under control.
The treatment reduces the number of drugs patients need to take. The April 21 approval also offers doctors an alternative option for people who may not tolerate other medications very well, Merck said.
Islatravir is an experimental treatment; however, Merck’s doravirine received approval in 2019. It sells in the U.S. under the brand name Pifeltro for HIV-1 treatment in combination with other antiretrovirals and as a single-tablet regimen.
The FDA approved Idvynso as a new HIV-1 therapy for adults who are virologically suppressed and have no history of virologic treatment failure or medication substitutions due to doravirine resistance.
HIV-1 is the most common strain of the retrovirus that causes acquired immunodeficiency syndrome, commonly known as AIDS. About 40.8 million people are living with HIV globally, and about 1.3 million new infections occur annually, according to the U.S. Centers for Disease Control and Prevention.
HIV statistics
- About 40.8M people are living with HIV globally.
- About 1.3M new infections occur annually.
SOURCE: CDC
During two late-stage studies of more than 1,00 patients, Idvynso showed strong HIV-1 suppression and was found to be as effective as existing standard three-drug therapies in adults.
A growing HIV portfolio
Merck Research Laboratories Chief Medical Officer and Senior Vice President Dr. Eliav Barr commented, “As the only two-drug, non-INSTI, tenofovir-free regimen, Idvynso expands therapeutic diversity beyond the currently available oral treatment options. As the health needs of adults living with HIV change over time, Idvynso gives clinicians a new choice for HIV treatment.
“This approval marks an important new chapter in Merck’s long-standing commitment to research and discovery for people living with HIV.”
According to Merck, Idvynso will hit pharmacies May 11. Patients can get help navigating insurance coverage, understanding out-of-pocket costs and accessing copay assistance via the Merck Access program. For more information, patients and providers can call 1-877-709-4455 or visit the program website.
Merck considers itself a leader in HIV research since 1985.
With the patent cliff for its blockbuster cancer drug Keytruda creeping closer, Merck is relying on its future HIV sales as a key spoke of its infectious disease drug portfolio.
According to a recent presentation, Merck’s HIV portfolio could represent a $15 billion commercial opportunity by the mid-2030s. More broadly, the company expects new products across its pipeline to generate more than $70 billion in annual revenue by that point.